More Information
Submitted: February 25, 2026 | Accepted: March 10, 2026 | Published: March 11, 2026
Citation: Juchaux F, Deloche-Bensmaine C, Chagnoleau C, Kovylkina N, El Haddad G, Guerin L, et al. Unveiling the Anti-aging Potential of a mix of Grifola frondosa, Peptides, and Rhamnose: Epidermal and Dermal Rejuvenation Performance. An In vitro Comparison with Retinol, Retinoic Acid and Vitamin C. Ann Dermatol Res. 2026; 10(1): 004-013. Available from:
https://dx.doi.org/10.29328/journal.adr.1001039
DOI: 10.29328/journal.adr.1001039
Copyright license: © 2026 Juchaux F, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: Collagens; Retinoid; Grifola frondosa; Peptides; Rhamnose
Unveiling the Anti-aging Potential of a mix of Grifola frondosa, Peptides, and Rhamnose: Epidermal and Dermal Rejuvenation Performance. An In vitro Comparison with Retinol, Retinoic Acid and Vitamin C
Franck Juchaux1* , Claire Deloche-Bensmaine2, Corinne Chagnoleau2, Natalia Kovylkina2, Georges El Haddad2, Lucie Guerin1 and Elisa Caberlotto2
, Claire Deloche-Bensmaine2, Corinne Chagnoleau2, Natalia Kovylkina2, Georges El Haddad2, Lucie Guerin1 and Elisa Caberlotto2
1L’Oreal Research and Innovation, Chevilly-Larue, France
2Vichy Laboratoires, Levallois-Perret, France
*Corresponding author: Franck Juchaux, L’Oréal Research and Innovation, Chevilly-Larue, France, Email: [email protected]
Skin aging is a complex and multifactorial phenomenon. While retinoids and vitamin C are considered gold standards in anti-aging, their side effects can be problematic. In this context, we have developed a novel combination of active ingredients: maitake, a peptide complex, and rhamnose, designed to improve the signs of aging by primarily targeting collagen. This study aimed to compare, in vitro, the effects of our new combination with those of retinol, retinoic acid, and vitamin C.
The different treatments were evaluated in a human skin model reconstituted in vitro from aged cells by measuring epidermal thickness and the expression of key biomarkers in the epidermis, the dermo-epidermal junction (DEJ), and the dermis. The efficacy of each treatment was then compared using a scoring system.
Our combination of active ingredients significantly reversed the aged phenotype by restoring epidermal thickness and stimulating hyaluronic acid production as well as the expression of key proteins such as DSG-1 and Collagens I, IV, V, VI, and VI. This mix demonstrated overall performance comparable to that of retinol, but superior to that of retinoic acid and vitamin C.
These results show that this new combination is a promising anti-aging solution, offering comprehensive action and potentially improved tolerability. In vivo clinical studies will be necessary to confirm these promising in vitro results.
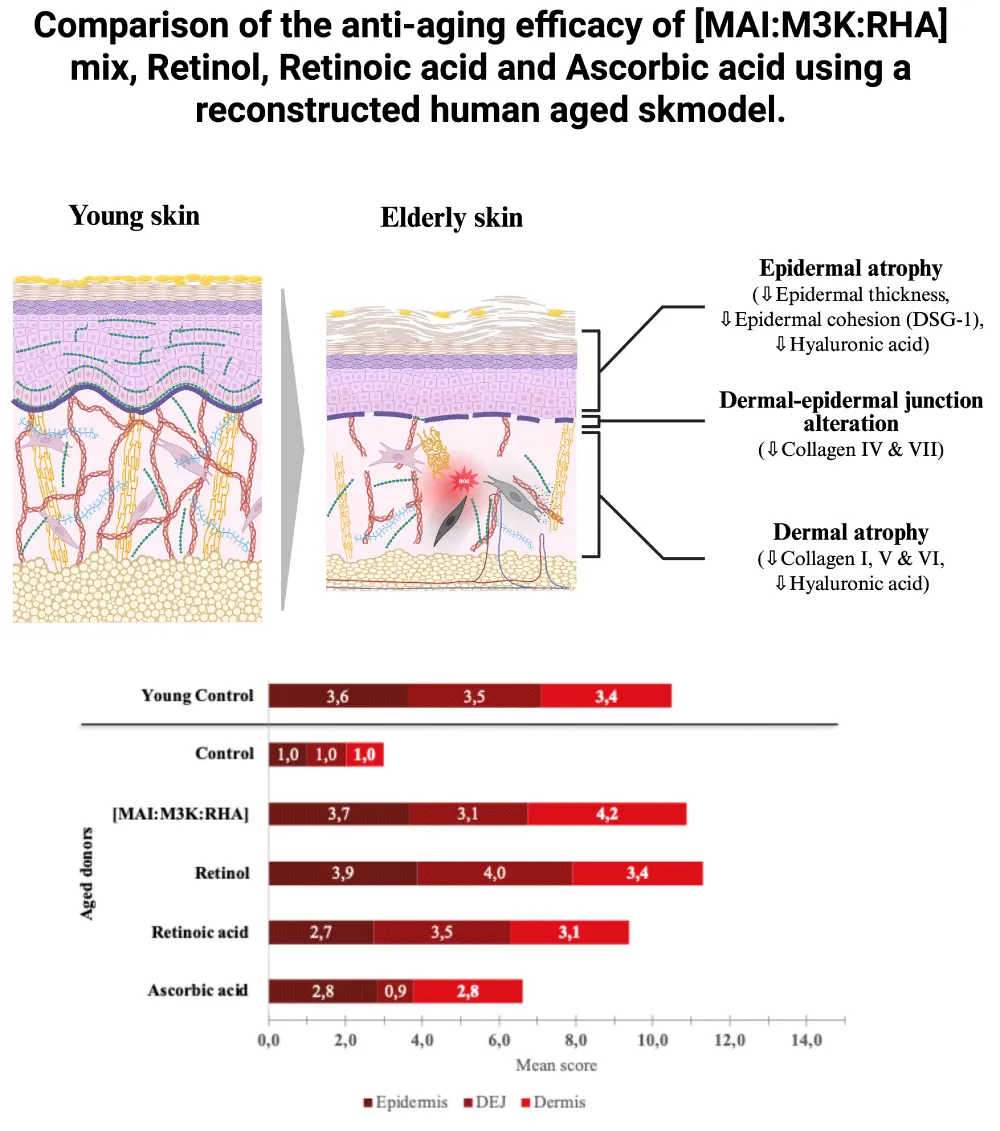
Graphical abstract: This study evaluates and compares the anti-aging efficacy of [MAI: M3K: RHA] mix, Retinol, Retinoic acid and Ascorbic acid in a reconstructed human aged skin by measuring their capacity to reverse the aged phenotype.
Graphical abstract: This study evaluates and compares the anti-aging efficacy of [MAI: M3K: RHA] mix, Retinol, Retinoic acid and Ascorbic acid in a reconstructed human aged skin by measuring their capacity to reverse the aged phenotype.
Skin aging is a complex and multifactorial phenomenon, influenced by both intrinsic chronological factors and extrinsic environmental stressors, such as ultraviolet (UV) radiation, pollution, and hormonal variations. The cumulative effect of these factors leads to significant morphological and functional alterations in the skin. Among the main changes are impaired barrier function, decreased epidermal thickness, flattening and disorganisation of the dermo-epidermal junction (DEJ), and marked dermal atrophy, characterised by the degradation of key components of the extracellular matrix (ECM), including collagen, elastic fibres, and glycosaminoglycans. These transformations collectively result in a loss of skin volume in all compartments, manifesting on the surface as visible signs of aging, such as wrinkles, fine lines, and decreased firmness and elasticity [1-6].
These morphological changes are intrinsically linked to alterations in central biological pathways, which profoundly impact cellular function and are widely recognised as a hallmark of aging [7]. Among the fourteen hallmarks identified to date, those related to changes in the extracellular matrix, and more specifically to alterations in collagen, have generated considerable interest. Collagen is the most abundant protein in the skin, constituting approximately 75 to 80% of its dry weight [8]. It is well documented that the collagen fibre network undergoes significant changes during aging, directly contributing to the appearance of visible signs of aging [9-11]. While numerous active ingredients are currently described for their ability to improve the signs of aging, retinoids and vitamin C are widely considered the gold standard in anti-aging dermatology [12,13].
Retinoids, such as retinoic acid and retinol, exert their effects primarily through the activation of RAR/RXR nuclear receptors. This activation regulates gene transcription, thereby influencing cell differentiation and various skin functions. Retinoids, in particular, promote epidermal thickening via the activation of the C-Jun transcription factor. Furthermore, they stimulate collagen synthesis by inducing the TGF-β/CTGF signalling pathway and limit its degradation by inhibiting, among other things, the expression of CCN1 and MMP1. These molecular effects underlie the anti-aging benefits observed with retinoid use. Although the anti-aging benefits of retinoids are widely described and recognised, their use is frequently associated with undesirable side effects such as irritation, peeling, burning sensations, and dermatitis. These adverse reactions can compromise patient acceptance of the treatment and limit its widespread use [14].
Vitamin C, an essential nutrient, acts as a powerful natural antioxidant, protecting cells from oxidative stress induced by free radicals. In addition to its antioxidant properties, vitamin C is a crucial cofactor for many enzymes involved in collagen synthesis, epidermal lipid synthesis, and pigmentation processes. While the effects of vitamin C are well documented in vitro, its efficacy in vivo, particularly when applied alone topically, is less well described. Vitamin C is often formulated in combination with other active ingredients, which complicates the interpretation of its isolated anti-aging benefits. Nevertheless, it remains a key ingredient in anti-aging skincare [15,16].
Although retinoids and vitamin C also target other biological aspects, collagen plays a central role. We hypothesized that preserving and restoring the collagen network is important for effectively correcting the visible signs of aging. In this context, we developed a novel combination of three active ingredients: maitake (MAI, extracted from Grifola frondosa Grey), a peptide complex (Matrixyl-3000®, Sederma France, hereafter M3K), and rhamnose (RHA). We previously demonstrated the efficacy of this combination ([MAI: M3K: RHA]) in modulating collagen diversity and strengthening the network by acting on both the quantity and quality of collagen fibres. Furthermore, we showed that this active combination can reverse the signs of skin aging [17]. Following its incorporation into a serum, we observed a significant improvement in 16 signs of aging after three months of treatment [18].
Our goal now is to compare the efficacy of our combination of active ingredients with standard dermatological treatments, namely retinoic acid, retinol, and vitamin C. Before conducting a comprehensive clinical study to evaluate and compare the effects of these treatments on the visible signs of aging, it seemed relevant to assess and compare them in vitro. To do this, we used a reconstituted human skin model, developed from aged cells, capable of reproducing the phenotype observed in mature skin. After preliminary cytotoxicity tests, all raw materials were systematically tested at their maximum non-cytotoxic concentrations. The efficacy of the treatments was evaluated by measuring morphological parameters, such as epidermal thickness, and by quantifying the expression of key biomarkers involved in the skin aging process, such as strengthening of the skin barrier function, strengthening of the dermo-epidermal junction, and strengthening of the dermis.
This article presents compelling evidence of the high efficacy of our new active complex compared to established dermatological actives and supports the interest in targeting collagens to combat the effects of aging.
Culture and treatments
Primary cultures of human fibroblasts and keratinocytes were established using healthy skin biopsy samples obtained from aged donors (age: 57 and 42 years, respectively), as previously described [19]. Human skin tissue was collected according to the principles of the Declaration of Helsinki, and its use was declared to the French Research Ministry. The donors provided written informed consent according to the 2014 French bioethics law (law number: 94–954 of July 29, 1994).
A dermis equivalent was reconstructed by seeding human aged dermal fibroblasts in a scaffold composed of collagen, glycosaminoglycans, and chitosan (LabSkin matriX®, LabSkin Creations, Lyon, France). After 10 days of culture Under optimized cell culture conditions for ECM neo-synthesis, as previously described [20], the medium was changed and replaced by a culture medium containing or not (control), our mix [MAI: M3K: RHA] (0.025%w/v MAI + 0.15%w/v M3K + 0.25%w/v RHA), or Retinoic acid (0.001 µM) or Retinol (0.01 µM) or Vitamin C (0.01%w/v). Dermis equivalents were cultured for an additional 11 days with renewal of medium and treatments every 2-3 days.
At the end of the incubation period, the culture medium was changed. Keratinocytes from an aged donor were seeded on the top of each dermis equivalent, and the tissues were cultured for 1 day (without actives). The culture medium was changed, treatments were applied as described previously, and tissues were cultured in immersion for 7 days with a renewal of culture medium and treatment every 2 days. The culture medium was renewed, tissues were placed at the air/liquid interface, and cultured for 7 days in the absence of treatment. At the end of the incubation period, the culture medium was replaced, the treatments were applied systemically as described previously, and the tissues were cultured for an additional 7 days. Reconstructed tissues with cells from young donors (age: 5 years) were cultured in parallel and used as a control, and all conditions were performed in triplicate.
At the end of the incubation, the tissues were fixed and embedded in paraffin for histological and immunohistological analysis.
Histological analysis and epidermal thickness measurement
To evaluate the global cutaneous structure of samples, Haematoxylin-phloxin-saffron (HPS) staining was performed. Paraffin sections of 5 μm of each condition were cut. After dewaxing and rehydration, the samples were stained with HPS. After rinsing, the sections were dehydrated before the mounting of the slides with a hydrophobic mounting medium. Epidermal thickness was obtained with a Euclidean distance map. Pixels corresponding to the epidermis were selected from other pixels. Images were converted to an 8-bit binary image. Images corresponding to the area of interest were converted to a 16-bit distance map. To each epidermis pixel (nonzero) in the distance map binary image, a value equal to its distance from the nearest background pixel (zero) was assigned. The epidermis basal line was selected and then applied to the distance map. The mean intensity of the basal line corresponds to the mean distance between the basal line and the stratum corneum. Data are expressed in μm. For all data, the statistical significance was assessed by running a one-way Student's test, and statistically significant differences are indicated by asterisks as follows: *p < 0.05, ** p < 0.01, and *** p < 0.001.
In situ immunolabeling and image analysis
We analysed paraffin-embedded sections using immunohistochemistry (Collagen I and Hyaluronic acid) or using immunofluorescence (Desmoglein-1, Collagen IV, Collagen VII, Collagen V, and Collagen VI). Following heat-mediated antigen retrieval treatment (incubation for 15 min at 95 °C in citrate buffer [Dako, Glostrup, Denmark]), phosphate-buffered saline containing 4% of bovine serum albumin was used to block non-specific binding. Subsequently, the sections were incubated with the primary antibody of interest (anti-collagen I, anti-collagen V, and anti-collagen VI from Novotec, Bron, France; anti-collagen IV from Dako, Glostrup, Denmark; anti-collagen VII, anti-hyaluronic acid, and anti-desmoglein-1 from Santa Cruz Biotechnology, Santa Cruz) diluted in phosphate-buffered saline containing 4% of bovine serum albumin overnight at room temperature. After incubation, for immunohistochemistry analysis, sections were incubated for 1 h with EnVisionTM anti-mouse/rabbit-HRP secondary antibody (EnVision+ System-HRP; Dako), and DAB+ substrate solution was applied to the sections to reveal the colour of the antibody staining. Next, slides were counterstained by immersing them in 25% Harris Hematoxylin counterstaining solution. As a negative control, incubation with the primary antibody was replaced by incubation with the corresponding immunoglobulin G class. For immunofluorescence analysis, after incubation, the sections were incubated for 1 h with an AlexaFluor-568-conjugated anti-mouse/rabbit secondary antibody (Molecular Probes, Invitrogen, Carlsbad, CA, USA) and subjected to nuclear counterstaining using 4′,6-diamidino-2-phenylindole. As a negative control, we prepared a condition in which the primary antibody was replaced by the corresponding immunoglobulin G class.
Immunostained specimens were observed using a Zeiss Axio Observer D1 microscope (Zeiss, Oberkochen, Germany). For the markers of interest, positively red-stained tissue areas were automatically detected and segmented from other pixels. The surface area of interest was measured automatically. Data were normalised by the DEJ length and dermal area for dermal-epidermal markers and dermal markers, respectively, and are expressed as a percentage of density.
Data analysis
Efficacy percentage calculation: To allow for long-term comparison across different studies, data were doubly normalised with respect to the effects observed in both the aged control and the young control, according to the formula:
In this way, an efficacy value of 100% corresponds to an effect identical to that observed in young skin.
Score calculation: A 6-point scoring scale has been implemented where a score of 1 corresponds to 0% efficacy, and a score of 5 corresponds to the maximum efficacy observable for a given biomarker. To ensure a linear scale, the interval between each score is equal to . Consequently, a treatment's score is calculated using the formula:
Statistical analysis
Statistical analysis was performed using a one-way ANOVA with Tukey's multiple comparisons test. Statistically significant differences are indicated by asterisks as follows:
*p < 0.05, **p < 0.01, ***p < 0.001, and ****p < 0.0001.
Effect on the epidermal compartment
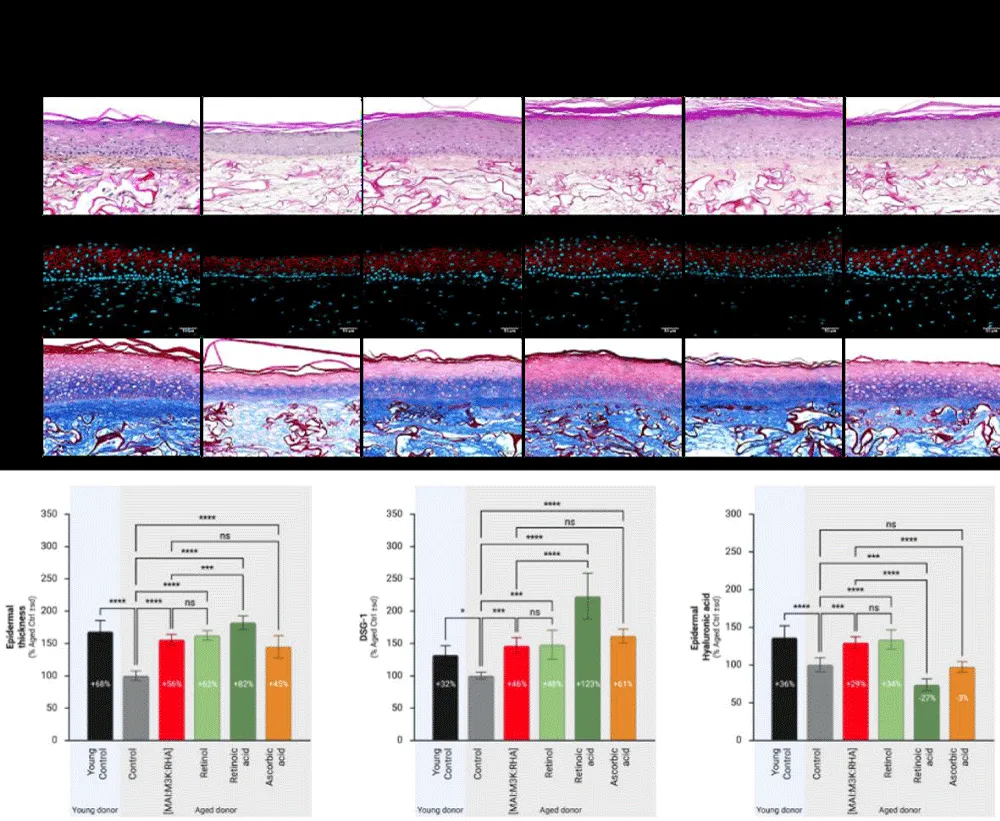
The epidermis reconstructed with cells from aged donors showed significant morphological alterations compared to that reconstructed with cells from young donors. In comparison to aged epidermis, young epidermis was thicker (+68% ± 17, p < 0.0001; Figure 1), more cohesive with increased Desmoglein-1 expression (+32% ± 14, p < 0.05; Figure 1), and also exhibited a higher level of hyaluronic acid expression (+36% ± 15, p < 0.0001; Figure 1).
Figure 1: Effect of [MAI: M3K: RHA] mix, Retinol, Retinoic acid, and Ascorbic acid on epidermal structure of human aged-reconstructed skins – Histological changes and biomarkers expression. Human aged-reconstructed skins were treated for a total of 35 days with [MAI:M3K: RHA] mix (0.025%p/v MAI + 0.15%p/v M3K + 0.25%p/v RHA) or Retinol at 0.1 µM or Retinoic acid at 0.001 µM or at 0.01%p/v. Reconstructed tissues with cells from young donors were cultured in parallel and used as a control. Epidermal thickness was measured by image analysis of histological staining. The expression of Desmoglein-3 and Hyaluronic acid was measured by image analysis after immunostaining. Abbreviations: MAI, Maitake; M3K, Palmitoyl Tripeptide-1: Palmitoyl Tetrapeptide-7; RHA, Rhamnose; HPS, Hematoxylin-phloxine-saffron; sd, standard deviation; *p < 0,05; **p < 0,01; ***p < 0,001; ****p < 0,0001.
[MAI: M3K: RHA] mix improved epidermal thickness of aged donors (+56% ± 8, p < 0.0001; Figure 1) as well as the expression of Desmogelin-1 (+46% ± 12, p < 0.001; Figure 1) and Hyaluronic acid (+29% ± 8, p < 0.001; Figure 1). Retinol improved epidermal thickness of aged donors (+62% ± 7, p < 0.0001; Figure 1) as well as the expression of Desmogelin-1 (+48% ± 23, p < 0.001; Figure 1) and Hyaluronic acid (+34% ± 13, p < 0.0001; Figure 1). Retinoic acid improved epidermal thickness of aged donors (+82% ± 10, p < 0.0001; Figure 1) as well as the expression of Desmogelin-1 (+123% ± 36, p < 0.0001; Figure 1) but not that of Hyaluronic acid, which was, on the contrary, inhibited (-27% ± 8, p < 0.001; Figure 1).
Finally, Ascorbic acid improved epidermal thickness of aged donors (+45% ± 17, p < 0.0001; Figure 1) as well as the expression of Desmogelin-1 (+61% ± 11, p < 0.0001; Figure 1) without modulating Hyaluronic acid expression -3% ± 7, ns; Figure 1).
The effect of [MAI: M3K: RHA] mix was on par with Retinol regarding the effects on epidermal thickening, Desmoglein-1, and Hyaluronic acid expression, but higher than that of Retinoic acid and Ascorbic acid. If the effect of Retinoic on epidermal thickness and Desmoglein-1 expression was clearly higher to that of Co-bonding technology (+82% ± 10 vs. +56% ± 8, p < 0.001and +123% ± 36 vs. +46% ± 12, p < 0.0001 respectively; Figure 1) it was clearly less on Hyaluronic acid expression (-27% ± 8 vs. +29% ± 8, p < 0.0001; Figure 1). For ascorbic acid, even if its effect was on par with that of Co-bonding technology on epidermal thickness and Desmoglein-1 expression, it was less on Hyaluronic acid expression (-3% ± 7 vs. +29% ± 8, p < 0.0001; Figure 1).
Effect on the dermal-epidermal junction
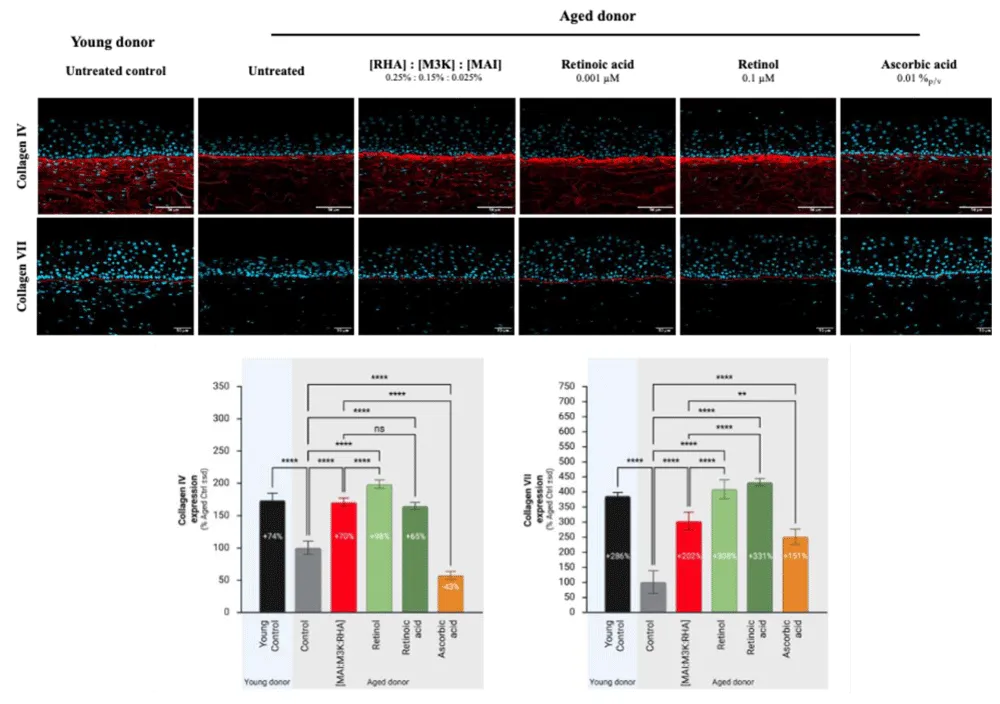
The quality of the dermal-epidermal junction was evaluated by measuring the expression of Collagen IV and Collagen VII. The expression of collagens IV and VII in tissues reconstructed with cells from young donors was clearly higher compared to that measured in tissues reconstructed with cells from aged donors (+74% ± 11, p < 0.0001 and +286% ± 11, p < 0.0001 repectively; Figure 2). This loss of expression of Collagens IV and VII with age is consistent with aging-induced dermal-epidermal junction alterations.
[MAI: M3K: RHA] mix stimulated the expression of both Collagens IV and VII (+70% 7, p < 0.0001; +202% ± 30, p < 0.0001, respectively; Figure 1). A stimulating effect on both markers was also observed with Retinol (+98% ± 7, p < 0.0001; +308% ± 31, p < 0.0001 respectively; Figure 1) and Retinoic acid (+65% ± 6, p < 0.0001; +331% ± 30, p < 0.0001 respectively; Figure 1). Ascorbic acid had a more contrasting effect, as it stimulated the expression of Collagen VII (+151% ± 25, p < 0.0001; Figure 2), whereas for Collagen IV, an inhibition was observed (-43% ± 6, p < 0.0001; Figure 2).
The effect of [MAI: M3K: RHA] mix on the improvement of dermal-epidermal junction was significantly higher than that of Ascorbic acid but significantly less than that of Retinol. In comparison with Retinoic acid, while the [MAI: M3K: RHA] mix was on par regarding the expression of Collagen IV, its effect was less concerning Collagen VII expression (Figure 2).
Figure 2: Effect of [MAI: M3K: RHA] mix, Retinol, Retinoic acid, and Ascorbic acid on the structure of dermal-epidermal junction in human aged-reconstructed skins – Histological changes and biomarkers expression. Human aged-reconstructed skins were treated for a total of 35 days with [MAI:M3K: RHA] mix (0.025%p/v MAI + 0.15%p/v M3K + 0.25%p/v RHA) or Retinol at 0.1 µM or Retinoic acid at 0.001 µM or at 0.01%p/v. Reconstructed tissues with cells from young donors were cultured in parallel and used as a control. The expression of Collagens IV and VII was measured by image analysis after immunostaining. Abbreviations: MAI, Maitake; M3K, Palmitoyl Tripeptide-1: Palmitoyl Tetrapeptide-7; RHA, Rhamnose; sd, standard deviation; *p < 0,05; **p < 0,01; ***p < 0,001; ****p < 0,0001.
Effect on dermis
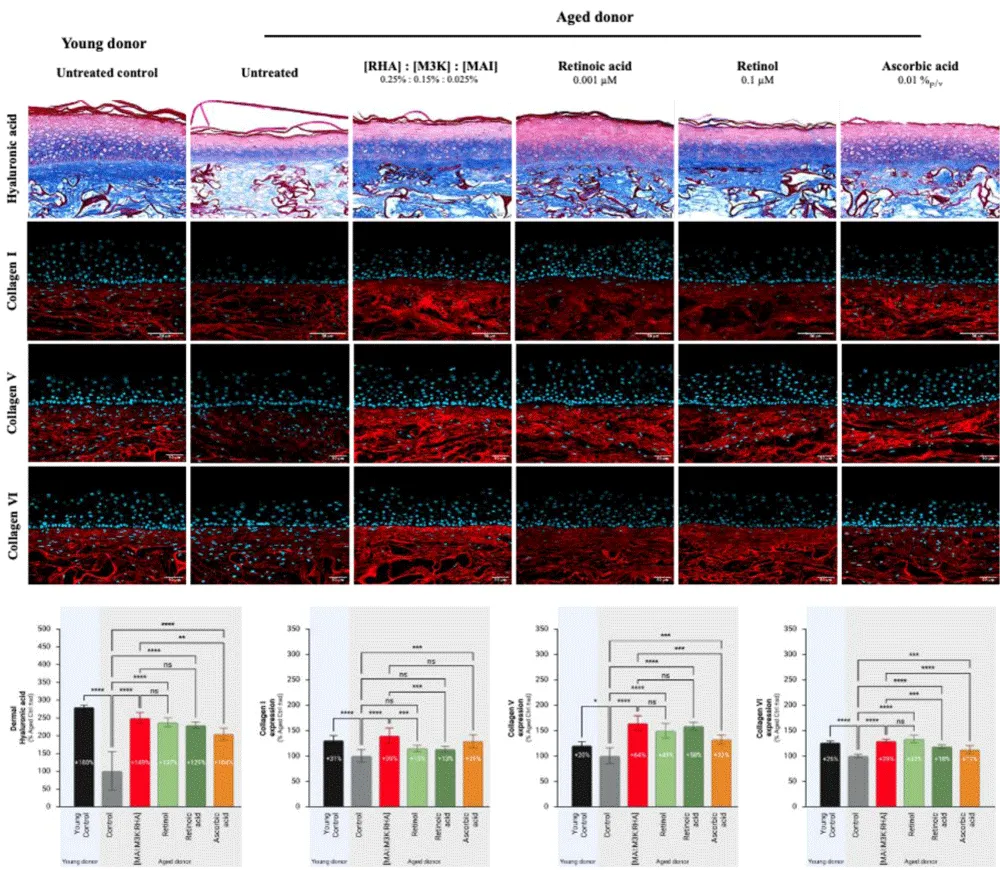
The quality and the structure of the dermis were evaluated by measuring the expression of Collagen I, V, and VI and by measuring the amount of Hyaluronic acid. In aged tissues, a clear decrease in the expression of all markers was observed compared to young tissues, where the expression of collagens I, V, and VI was significantly higher (+31% ± 9, p < 0.0001; +20% ± 8, p < 0.05 and +26% ± 3, p < 0.0001repectively; Figure 3). Similarly, the HA content in young skin was also higher compared to aged skin (+180% ± 6, p < 0,0001; Figure 3).
This loss of expression of collagens in the dermis, as well as the loss of HA, is consistent with aging-induced dermal atrophy.
[MAI: M3K: RHA] mix stimulated the expression of all Collagens I, V and VI (+39% ± 15, p < 0.0001; +64% ± 15, p < 0.0001 and + 29% ± 4, p < 0.0001respectively; Figure 3) as well as the expression of dermal Hyaluronic acid (149% ± 15, p < 0.0001; Figure 3). Retinol stimulated the expression of Collagens V, VI, and Hyaluronic acid (+49% ± 14, p < 0.0001, +33% ± 7, p < 0.0001, and +137% ± 13, p < 0.0001, respectively; Figure 3) but not that of Collagen I (+15% ± 6, ns; Figure 3). A similar effect was observed with Retinoic acid, where only a stimulation of Collagens V, VI, and Hyaluronic acid was observed (+58% 8, p < 0.0001, +18% ± 4, p < 0.0001, and +129% ± 9, p < 0.0001, respectively; Figure 3). Finally, Ascorbic acid stimulated the expression of Collagens I, V, and VI (+29% ± 13, p < 0.001, +32% ± 9, p < 0.001, and +12% ± 8, p < 0.001, respectively; Figure 3) as well as the expression of Hyaluronic acid (+104% ± 17, ns; Figure 3).
Figure 3: Effect of [MAI:M3K: RHA] mix, Retinol, Retinoic acid, and Ascorbic acid on the expression of dermal components in human aged-reconstructed skins – Histological changes and biomarkers expression. Human aged-reconstructed skins were treated for a total of 35 days with [MAI:M3K: RHA] mix or Retinol at 0.1 µM or Retinoic acid at 0.001 µM or at 0.01%p/v. Reconstructed tissues with cells from young donors were cultured in parallel and used as a control. The expression of Collagens I, V, and VI was measured by image analysis after immunostaining. Abbreviations: MAI, Maitake; M3K, Palmitoyl Tripeptide-1: Palmitoyl Tetrapeptide-7; RHA, Rhamnose; sd, standard deviation; *p < 0,05; **p < 0,01; ***p < 0,001; ****p < 0,0001.
The effect of [MAI: M3K: RHA] mix on the improvement of dermis was superior to that of Retinol, Retinoic acid, and Ascorbic acid. Although retinoids were on par with [MAI:M3K: RHA] mix on the expression of Collagen V, Collagen VI, and Hyaluronic acid, their effect was less potent on Collagen I expression (+15% and +13%, respectively, versus +39%, p < 0.001). For ascorbic acid, although its effect was not statistically inferior for Collagen I, it was indeed inferior for Collagen V, Collagen VI, and Hyaluronic acid (+32% vs. +64% and +13% vs. +29% respectively, p < 0.001 and +104% vs. +149%, p < 0.01).
Overall anti-aging efficacy
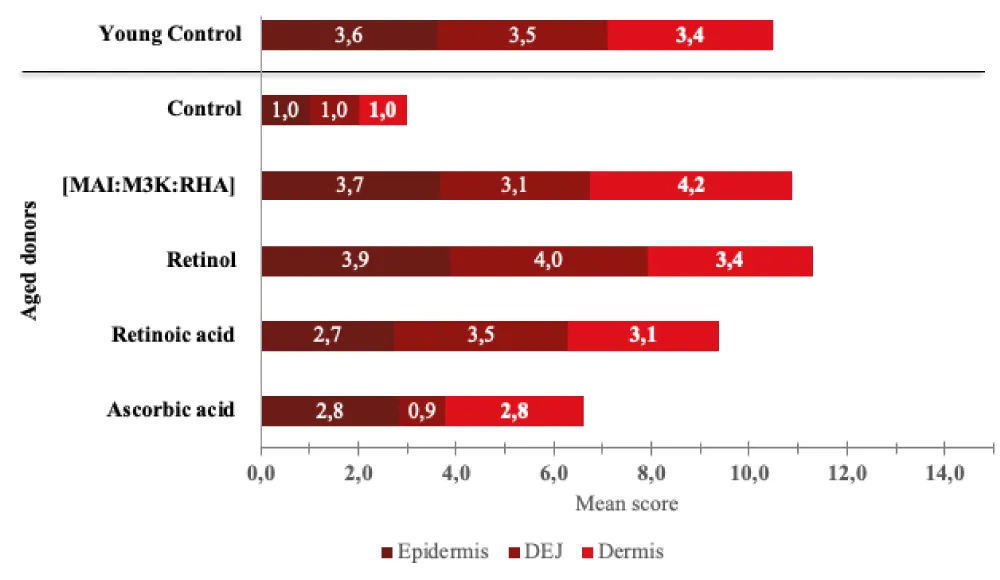
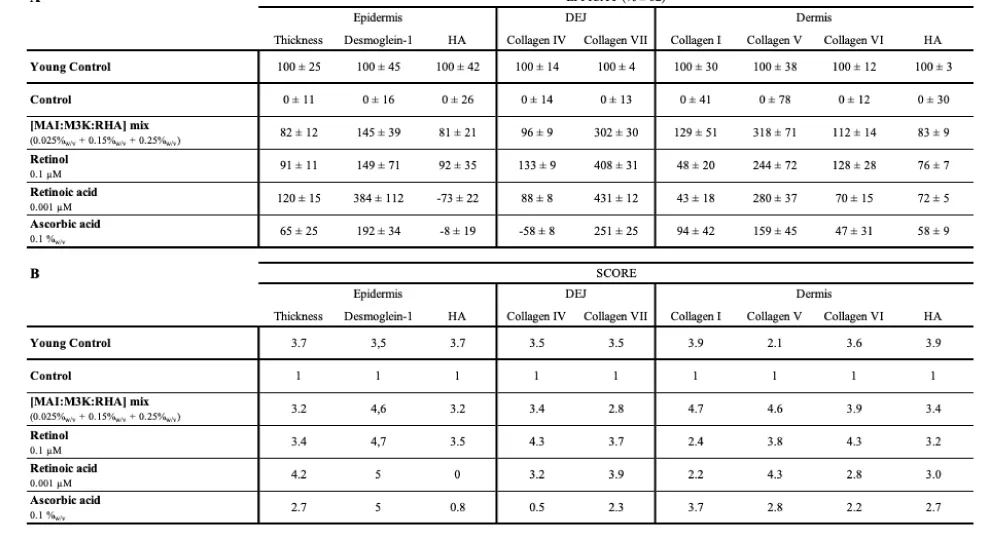
In order to compare the overall anti-aging efficacy of the treatments, an efficacy percentage, taking into account the observed differences between young and aged donor tissues, was calculated (Table 1A), and a 5-point score was calculated (Table 1B). This allowed us to derive an average score for each of the epidermal, DEJ (Dermo-Epidermal Junction), and dermal compartments (Figure 4). These results show that [MAI: M3K: RHA] mix and Retinol are on par and exhibit the best anti-aging efficacy. While Retinol showed the best efficacy at the DEJ level (score 4 vs. 3.1), in the dermis, [MAI: M3K: RHA] mix demonstrated the highest efficacy (score 3.4 vs. 4.2). Retinoic acid, on the other hand, demonstrated strong efficacy in the epidermis, although no clear effect was observed on HA expression. A clear effect was also observed at the dermo-epidermal junction, while a lesser effect was observed in the dermis. Finally, ascorbic acid generally showed weaker effects on all three compartments (Figure 4).
Figure 4: Global anti-aging efficacy of [MAI: M3K: RHA] mix, Retinol, Retinoic acid, and Ascorbic acid – Meta-analysis.
Table 1: Global anti-aging efficacy of [MAI: M3K: RHA] mix, Retinol, Retinoic acid, and Ascorbic acid – Meta-analysis. An efficacy percentage, taking into account the observed differences between young and aged donor tissue, was calculated (A), and a 5-point score was calculated (B).
The primary objective of this study was to evaluate the biological anti-aging efficacy of [MAI: M3K: RHA] mix and compare it with gold standards anti§aging dermatological actives, namely Retinol, Retinoic Acid, and Vitamin C. Specifically, our focus was on addressing structural skin alterations associated with aging, such as the appearance of wrinkles, fine lines, and loss of firmness. While these three active compounds exhibit distinct mechanisms of action, they converge on the common goal of restoring skin structure. To achieve this, we used an aged-reconstructed skin model, which accurately replicates major alterations of an aged phenotype by incorporating cells from elderly donors. This approach, combining morphological analysis and quantification of various biomarkers, allows for the detailed evaluation and comparison of the effects of different treatments.
First, we demonstrated the relevance of the aged-reconstructed skin model, reconstructed with cells from an elderly donor, by comparing it to a similar model reconstructed with cells from a young donor. Aged tissues displayed a thinner epidermis and decreased Desmoglein-1 expression, indicating a reduced epidermal turnover and an impaired barrier function. Epidermal hyaluronic acid (HA) content was also decreased. These observations are consistent with the phenotype observed in aged skin. It is well known that epidermal thickness decreases with age [21], and skin barrier function is compromised due to the alteration of the differentiation process, including the production of tight junction proteins, such as Desmoglein [22]. Furthermore, the loss of epidermal HA is considered a hallmark of cutaneous senescence [23]. At the dermal-epidermal junction (DEJ) level, assessing flattening in reconstructed models currently remains impossible due to the absence of natural undulations, even in young models. Therefore, to monitor DEJ evolution during aging, we quantified Collagen IV and Collagen VII expression, two key markers for DEJ integrity, and both are known to be altered within aging [24,25]. The significant decrease in the expression of these collagens in the aged model is consistent with physiological observations during aging. Finally, within the dermis, we assessed the expression of Collagens I, V, and VI, along with HA content. Collagen I is a key indicator of dermal aging [1-6], and Collagen V degeneration plays a significant role in aging due to its involvement in regulating collagen fibril diameter [26]. Collagen VI is a key regulator of dermal matrix assembly, composition, and fibroblast behaviour, potentially influencing wound healing and tissue regeneration, with its expression also being altered in photo-aged skin in response to M1/M2 macrophage skewing [27,28]. The loss of expression observed for these three types of collagens in reconstructed aged tissues is consistent with the establishment of an aged phenotype. A reduction in HA expression was also observed, in accordance with data from the literature [29].
In this study, we clearly demonstrated the benefits of our [MAI: M3K: RHA] mix across all skin compartments: epidermal, dermal-epidermal junction, and dermal. In the epidermis, treatment resulted in a clear increase of epidermal thickness, with an increase of DSG-1 and epidermal hyaluronic acid expression. After treatment, the epidermis exhibited a structure and morphology similar to that observed in tissues reconstructed with young donor cells. These results indicate a stimulation of epidermal renewal and a strengthening of the differentiation process and barrier function. A significant increase in the expression of collagens IV and VII was also observed, indicating a reinforcement of the dermo-epidermal junction (DEJ). The level of collagen IV expression was equivalent to that observed in reconstructed young skin, while the level of collagen VII was significantly higher. These results demonstrate thus the potential of our [MAI: M3K: RHA] mixture for strengthening the DEJ. Furthermore, in the dermis, all evaluated markers (collagen I, collagen V, collagen VI, and hyaluronic acid) were stimulated, further demonstrating the value of this treatment in combating age-related dermal atrophy. These results replicate those of a previous study where we demonstrated the ability of the [MAI: M3K: RHA] mix to stimulate all collagen families and help the skin to regain its youthful structure [17]. These results also help to explain the effects observed clinically after this combination was incorporated into a serum. Indeed, an improvement in the visible signs of aging, as well as an increase in collagen density, was observed [18]. This study also provides new insights into our [MAI: M3K: RHA] mix, as we did not anticipate a positive effect on hyaluronic acid (HA) synthesis, given that its constituents are not generally known to stimulate HA production. Although rhamnose is known to inhibit hyaluronidases [30], age-related HA loss is primarily due to decreased synthesis rather than increased degradation [29].
Retinoids, including Retinol and Retinoic Acid, also exhibited anti-aging effects across epidermal, DEJ, and dermal compartments. However, distinct differences were noted between these two molecules. At the epidermal level, retinol induced a marked increase in epidermal thickness, DSG-1 expression, and HA synthesis, consistent with what is widely described in the literature [31]. In contrast, although the effect of retinoic acid on epidermal thickening and DSG-1 expression was greater than that of retinol, no effect on HA synthesis was observed, contradicting the data in the literature. Nevertheless, despite its superior efficacy compared to retinol, retinoic acid is also associated with poorer tolerability. In in vitro tests, these problems often manifest as increased cytotoxicity, and the doses evaluated are generally lower than those used clinically. The lack of effect on HA synthesis in the epidermis could be attributed to an insufficient concentration of retinoic acid to stimulate HA production. At the dermo-epidermal junction (DEJ), retinol and retinoic acid demonstrated comparable effects in stimulating the expression of collagens IV and VII, consistent with data from the literature describing their role in improving the DEJ [32,33]. Finally, in the dermis, both retinol and retinoic acid stimulated the expression of collagens V and VI, as well as hyaluronic acid, without significantly stimulating that of collagen I. These effects are consistent with the anti-aging properties of retinoids. However, the lack of effect on collagen I expression remains surprising. However, it should be noted that most studies reporting effects on the expression of collagens I and III are based primarily on gene expression measurements [34]. Yet, what is predominantly described is the inhibitory effect of retinoids on MMP-1 expression in photoaged skin [35]. Thus, despite some differences observed in this study at the level of specific biomarkers, the results nevertheless demonstrate the strong anti-aging potential of retinoids.
Finally, Vitamin C also demonstrated an anti-aging efficacy by stimulating the expression of all biomarkers and enhancing epidermal turnover. Only hyaluronic acid was not induced, but there is no existing literature supporting an effect of Vitamin C on epidermal HA synthesis. These results are consistent with the established anti-aging effects of Vitamin C [15,16].
Beyond confirming the anti-aging effects of the different treatments, the main objective of this study was to compare them, but a marker-by-marker analysis proved too complex to establish a precise ranking of these different molecules. Therefore, we implemented an efficacy calculation, which takes into account the difference between young and aged control tissues, to quantify the ability of each treatment to reverse the aging phenotype. A five-point scoring system was then applied, and the average of these scores yielded an overall score for each compartment. This analysis shows that the [MAI: M3K: RHA] mix is comparable to retinol, although the latter obtained a lower dermal score. The main difference between the two treatments lies at the dermo-epidermal junction (DEJ), where retinol demonstrated a more pronounced effect. Furthermore, this analysis also highlighted a superior effect of both [MAI: M3K: RHA] mix and retinol compared to that of retinoic acid and vitamin C. It is important to note that this ranking is based on in vitro analysis and that in vivo confirmation, in a clinical study measuring the visible signs of aging, is necessary.
However, these results still demonstrate the potential of our [MAI: M3K: RHA] mix, even without considering tolerability. Indeed, despite their high efficacy, retinoids are often poorly tolerated, causing dryness, irritation, and redness, which hinders treatment adherence. An adjustment period is generally necessary to tolerate retinoids and continue with daily application. The good tolerability of our technology could therefore, in the long term, provide a clear advantage in terms of efficacy compared to retinoids. These results thus justify further comparisons within the framework of a clinical study.
This study clearly demonstrated the high anti-aging efficacy of our [MAI: M3K: RHA] mix, which improves all skin compartments in an aged-reconstructed skin model. The epidermis, the dermis, and the subcutaneous layer were all improved after treatment. Comparative analysis also revealed that its performance is equivalent to retinol and even superior to retinoic acid and vitamin C. These results highlight the potential of the [MAI: M3K: RHA] mix as a promising and well-tolerated innovative solution for combating skin aging, thus justifying further in vivo investigations.
We thank LabSkin Creations for its evaluation. We also thank our internal review committee for their help during the revision of this document.
Funding
Sponsorship for this study was funded by L’Oreal Research and Innovations.
Conflict of interest
Franck Juchaux, Claire Deloche-Bensmaine, Corinne Chagnoleau, Natalia Kovylkina, Georges El Haddad, Lucie Guérin and Elisa Caberlotto are employees of L’Oréal.
Author contributions
Conceptualization: FJ, CD; Methodology: FJ; Formal analysis and investigation: FJ; Writing – original draft preparation: FJ;
Writing – review and editing: CD, CC, NK, GE, LG, EC; Resources: CD and EC. All authors have read and approved the final manuscript.
Ethics approval
Human skin tissue was collected according to the principles of the Declaration of Helsinki of 1964 (and its subsequent amendments), and its use was declared to the French Research Ministry (declaration number: DC-2024-6232). The donors provided written informed consent according to the 2014 French bioethics law (law number: 94–954 of July 29, 1994).
- Naharro-Rodriguez J, Bacci S, Hernandez-Bule ML, Perez-Gonzalez A, Fernandez-Guarino M. Decoding skin aging: a review of mechanisms, markers, and modern therapies. Cosmetics. 2025;12:144. Available from: https://www.mdpi.com/2079-9284/12/4/144
- Wang Z, Man MQ, Li T, Elias PM, Mauro TM. Aging-associated alterations in epidermal function and their clinical significance. Aging (Albany NY). 2020;12(6):5551-5565. Available from: https://doi.org/10.18632/aging.102946
- Farage MA, Miller KW, Maibach HI. Degenerative changes in aging skin. In: Farage M, Miller K, Maibach H, editors. Textbook of aging skin. Berlin: Springer; 2017. Available from: https://link.springer.com/rwe/10.1007/978-3-662-47398-6_4
- Huang S, Strange A, Maeva A, Siddiqui S, Bastien P, Aguayo S, et al. Quantitative nanohistology of aging dermal collagen. Front Aging. 2023;4:1178566. Available from: https://doi.org/10.3389/fragi.2023.1178566
- Jevtić M, Löwa A, Nováčková A, Kováčik A, Kaessmeyer S, Erdmann G, et al. Impact of intercellular crosstalk between epidermal keratinocytes and dermal fibroblasts on skin homeostasis. Biochim Biophys Acta Mol Cell Res. 2020;1867(8):118722. Available from: https://doi.org/10.1016/j.bbamcr.2020.118722
- Coleman SR, Grover R. The anatomy of the aging face: volume loss and changes in 3-dimensional topography. Aesthet Surg J. 2006;26(1 Suppl):S4-9. Available from: https://doi.org/10.1016/j.asj.2005.09.012
- Kroemer G, Maier AB, Cuervo AM, Gladyshev VN, Ferrucci L, Gorbunova V, et al. From geroscience to precision geromedicine: understanding and managing aging. Cell. 2025;188(8):2043-2062. Available from: https://doi.org/10.1016/j.cell.2025.03.011
- Tzaphlidou M. The role of collagen and elastin in aged skin: an image processing approach. Micron. 2004;35(3):173-177. Available from: https://doi.org/10.1016/j.micron.2003.11.003
- Fligiel SE, Varani J, Datta SC, Kang S, Fisher GJ, Voorhees JJ. Collagen degradation in aged/photodamaged skin in vivo and after exposure to matrix metalloproteinase-1 in vitro. J Invest Dermatol. 2003;120(5):842-848. Available from: https://doi.org/10.1046/j.1523-1747.2003.12148.x
- Varani J, Warner RL, Gharaee-Kermani M, Phan SH, Kang S, Chung JH, et al. Vitamin A antagonises decreased cell growth and elevated collagen-degrading matrix metalloproteinases and stimulates collagen accumulation in naturally aged human skin. J Invest Dermatol. 2000;114(3):480-486. Available from: https://doi.org/10.1046/j.1523-1747.2000.00902.x
- Jansen KA, Licup AJ, Sharma A, Rens R, MacKintosh FC, Koenderink GH. The role of network architecture in collagen mechanics. Biophys J. 2018;114(11):2665-2678. Available from: https://doi.org/10.1016/j.bpj.2018.04.043
- Siddiqui Z, Zufall A, Nash M, Rao D, Hirani R, Russo M. Comparing tretinoin to other topical therapies in the treatment of skin photoaging: a systematic review. Am J Clin Dermatol. 2024;25(6):873-890. Available from: https://doi.org/10.1007/s40257-024-00893-w
- Jaros-Sajda A, Budzisz E, Erkiert-Polguj A. Ascorbic acid treatments as effective and safe anti-aging therapies for sensitive skin. Antioxidants (Basel). 2024;13(2):174. Available from: https://doi.org/10.3390/antiox13020174
- Quan T. Human skin aging and the anti-aging properties of retinol. Biomolecules. 2023;13:1614. Available from: https://doi.org/10.3390/biom13111614
- Pullar JM, Carr AC, Vissers MCM. The roles of vitamin C in skin health. Nutrients. 2017;9(8):866. Available from: https://doi.org/10.3390/nu9080866
- Traikovich SS. Use of topical ascorbic acid and its effects on photodamaged skin topography. Arch Otolaryngol Head Neck Surg. 1999;125(10):1091-1098. Available from: https://doi.org/10.1001/archotol.125.10.1091
- Juchaux F, Martinuzzi T, Deloche-Bensmaine C, Guerin L, Kovylkina N. A co-bonding technology combining maitake, a peptide complex, and rhamnose stimulates different skin collagens and reverses the age-phenotype: results from in vitro assays and a 3D reconstructed skin model. J Dermatol Cosmetol. 2025. Available from: https://medcraveonline.com/JDC/JDC-10-00310.pdf
- Juchaux F, Martinuzzi T, Guerin L, Simonnet JT, Kovylkina N, Martin E. Impact of targeting collagen diversity on skin aging signs: a pilot study. J Dermatol Cosmetol. 2025;9:37-40. Available from: https://doi.org/10.15406/jdc.2025.09.00288
- Germain L, Rouabhia M, Guignard R, Carrier L, Bouvard V, Auger FA. Improvement of human keratinocyte isolation and culture using thermolysin. Burns. 1993;19:99-104. Available from: https://doi.org/10.1016/0305-4179(93)90028-7
- Dos Santos M, Metral E, Boher A, Rousselle P, Thepot A, Damour O. An in vitro 3-D model based on extending the time of culture for studying chronological epidermis aging. Matrix Biol. 2015;47:85-97. Available from: https://doi.org/10.1016/j.matbio.2015.03.009
- Arnal-Forné M, Molina-García T, Ortega M, Marcos-Garcés V, Molina P, Ferrández-Izquierdo A, et al. Changes in human skin composition due to intrinsic aging: a histologic and morphometric study. Histochem Cell Biol. 2024;162:259-271. Available from: https://doi.org/10.1007/s00418-024-02305-w
- Lee AY. Molecular mechanism of epidermal barrier dysfunction as primary abnormalities. Int J Mol Sci. 2020;21:1194. Available from: https://doi.org/10.3390/ijms21041194
- Meyer LJ, Stern R. Age-dependent changes of hyaluronan in human skin. J Invest Dermatol. 1994;102(3):385-389. Available from: https://doi.org/10.1111/1523-1747.ep12371800
- Langton AK, Halai P, Griffiths CE, Sherratt MJ, Watson RE. The impact of intrinsic ageing on the protein composition of the dermal-epidermal junction. Mech Ageing Dev. 2016;156:14-16. Available from: https://doi.org/10.1016/j.mad.2016.03.006
- Bonnet I, Jobeili L, Cadau S, Berthélémy N, Pierrot A, Tedeschi C, et al. Collagen XVIII: a key interfacial component of the skin architecture. J Cosmet Sci. 2017;68(1):35-41. Available from: https://pubmed.ncbi.nlm.nih.gov/29465380/
- Jarrold BB, Tamura M, Bascom CC, Isfort RJ, Alora-Palli MB, Kimball AB. Collagen type V expression and localization in aging skin. J Am Acad Dermatol. 2017;76(6):AB75. Available from: https://www.em-consulte.com/article/1135159/collagen-type-v-expression-and-localization-in-agi
- Theocharidis G, Drymoussi Z, Kao AP, Barber AH, Lee DA, Braun KM, et al. Type VI collagen regulates dermal matrix assembly and fibroblast motility. J Invest Dermatol. 2016;136(1):74-83. Available from: https://doi.org/10.1038/jid.2015.352
- Horiba S, Kawamoto M, Tobita R, Kami R, Ogura Y, Hosoi J. M1/M2 macrophage skewing is related to reduction in types I, V, and VI collagens with aging in sun-exposed human skin. JID Innov. 2023;3(6):100222. Available from: https://doi.org/10.1016/j.xjidi.2023.100222
- Terazawa S, Nakajima H, Tobita K, Imokawa G. The decreased secretion of hyaluronan by older human fibroblasts under physiological conditions is mainly associated with the down-regulated expression of hyaluronan synthases but not with the expression levels of hyaluronidases. Cytotechnology. 2015;67(4):609-620. Available from: https://doi.org/10.1007/s10616-014-9707-2
- Novotná R, Škařupová D, Hanyk J, Ulrichová J, Křen V, Bojarová P, Brodsky K, Vostálová J, Franková J. Hesperidin, hesperetin, rutinose, and rhamnose act as skin anti-aging agents. Molecules. 2023;28(4):1728. Available from: https://doi.org/10.3390/molecules28041728
- Li WH, Wong HK, Serrano J, Randhawa M, Kaur S, Southall MD, Parsa R. Topical stabilized retinol treatment induces the expression of HAS genes and HA production in human skin in vitro and in vivo. Arch Dermatol Res. 2017;309(4):275-283. Available from: https://doi.org/10.1007/s00403-017-1723-6
- Bluemke A, Ring AP, Immeyer J, Hoff A, Eisenberg T, Gerwat W, Meyer F, Breitkreutz S, Klinger LM, Brandner JM, Sandig G, Seifert M, Segger D, Rippke F, Schweiger D. Multidirectional activity of bakuchiol against cellular mechanisms of facial ageing—experimental evidence for a holistic treatment approach. Int J Cosmet Sci. 2022;44(3):377-393. Available from: https://doi.org/10.1111/ics.12784
- Woodley DT, Zelickson AS, Briggaman RA, Hamilton TA, Weiss JS, Ellis CN, Voorhees JJ. Treatment of photoaged skin with topical tretinoin increases epidermal-dermal anchoring fibrils: a preliminary report. JAMA. 1990;263(22):3057-3059. Available from: https://pubmed.ncbi.nlm.nih.gov/2342217/
- Mukherjee S, Date A, Patravale V, Korting HC, Roeder A, Weindl G. Retinoids in the treatment of skin aging: an overview of clinical efficacy and safety. Clin Interv Aging. 2006;1(4):327-348. Available from: https://doi.org/10.2147/ciia.2006.1.4.327
- Varani J, Warner RL, Gharaee-Kermani M, Phan SH, Kang S, Chung JH, Wang ZQ, Datta SC, Fisher GJ, Voorhees JJ. Vitamin A antagonizes decreased cell growth and elevated collagen-degrading matrix metalloproteinases and stimulates collagen accumulation in naturally aged human skin. J Invest Dermatol. 2000;114(3):480-486. Available from: https://doi.org/10.1046/j.1523-1747.2000.00902.x